Adenosine Deaminase Severe Combined Immunodeficiency (ADA-SCID)
When you have a severe combined immunodeficiency (SCID), your immune system can’t fight off even mild infections on its own.With adenosine deaminase severe combined immunodeficiency (ADA-SCID), your body’s defenses stop working because of a problem with your genes. ADA-SCID is a serious disease that usually shows up early in life. Treatments can help, though, and people who get treatment before an infection happens can live long, healthy lives.
Everyone has ADA genes. If you have ADA deficiency, you have a glitch (mutation) in yours. As a result, your body doesn’t make enough of a certain tool, called an enzyme, that helps your white blood cells protect you from getting sick. Without that protection, you can easily get infections.
If your child is born with ADA deficiency, he’ll probably get a SCID diagnosis by the time he’s 6 months old. If the disease starts later, the symptoms may be less severe.
With treatment, you can manage symptoms and avoid infections. Left untreated, the body becomes less and less able to fight infections, which can be life-threatening.
Adenosine deaminase deficiency (ADA) is an inherited condition that affects the immune system and typically leads to severe combined immunodeficiency (SCID). People with SCID have a reduced or absent immune response which leaves them vulnerable to frequent bacterial, viral, and fungal infections. Most people affected by ADA develop symptoms of the condition before 6 months of age. However, approximately 10-15% of affected people have a “delayed” onset of symptoms; diagnosis of these cases, which are often less severe, typically takes place later in childhood (often between age 1 and 10) or even into adulthood. Signs and symptoms of ADA include pneumonia, chronic diarrhea, widespread skin rashes, slowed growth and/or developmental delay. ADA is caused by changes (mutations) in the ADA gene and is inherited in an autosomal recessive manner.
Patients of ADA-SCID have mutations on chromosome 20 as it is where the highly conservative ADA gene located. ADA-SCID can be treated by either stem cell transplant, or gene therapy or enzyme replacement therapy.
Causes
You get ADA-SCID only if both your parents pass on a copy of a faulty gene to you. If you get a copy from just one parent, you won’t get the disorder, but you can pass that copy on to your own children.
Adenosine deaminase deficiency is caused by changes (mutations) in the ADA gene. This gene encodes an enzyme that is found in the lymphocytes (specialized white blood cells), which are an important part of the immune system and help protect the body from infections. The function of this enzyme is to convert a substance that is harmful to lymphocytes (called deoxyadenosine) to a non-toxic material. Mutations in the ADA gene cause this enzyme to have reduced or absent function, allowing deoxyadenosine to buildup in the lymphocytes. This results in the loss of lymphocytes before they are able to mature and fight infection, leading to severe combined immunodeficiency.
The enzyme adenosine deaminase is encoded by a gene on chromosome 20. ADA deficiency is inherited in an autosomal recessive manner. This means the defective gene responsible for the disorder is located on an autosome (chromosome 20 is an autosome), and two copies of the defective gene (one inherited from each parent) are required in order to be born with the disorder. The parents of an individual with an autosomal recessive disorder both carry one copy of the defective gene, but usually do not experience any signs or symptoms of the disorder. Age of onset and severity is related to some 29 known genotypes associated with the disorder.
Adenosine deaminase deficiency is inherited in an autosomal recessive manner. This means that to be affected, a person must have a mutation in both copies of the responsible gene in each cell. The parents of an affected person usually each carry one mutated copy of the gene and are referred to as carriers. Carriers typically do not show signs or symptoms of the condition. When two carriers of an autosomal recessive condition have children, each child has a 25% (1 in 4) risk to have the condition, a 50% (1 in 2) risk to be a carrier like each of the parents, and a 25% chance to not have the condition and not be a carrier.
Signs and Symptoms
Symptoms usually appear in the first months of life. If your baby has this disease, he may have a lot of infections in different parts of his body, including:
- Ear
- Sinus
- Mouth
- Lung
- Skin
It’s common for babies to get infections. Pay special attention if your child has them often and they:
- Are severe
- Are long-lasting
- Are unusual
- Frequently recur
- Children with ADA-SCID often have diarrhea and widespread skin rashes. They may also grow slowly and have delayed progress in other development areas like motor and social skills.
If the disease doesn’t appear until later in childhood or adulthood, the symptoms may be mild at first. The first symptoms may be ear or upper-respiratory infections that keep coming back.
In most cases, signs and symptoms of adenosine deaminase deficiency (ADA) develop before 6 months of age. These affected babies are usually diagnosed with severe combined immunodeficiency (SCID), a condition characterized by a reduced or absent immune response. Babies with SCID are vulnerable to frequent bacterial, viral, and fungal infections. Although many of these infections would cause no illness or only mild disease in healthy children, they can be very severe and even life-threatening in children with SCID. Signs and symptoms of SCID include chronic diarrhea; pneumonia and other lung infections; widespread skin rashes; slowed growth; absent tonsils and lymph nodes; bone abnormalities; and/or developmental delay.
Approximately 10-15% of affected people have a “delayed” onset of symptoms, which typically develop later in childhood (often between age 1 and 10) or even into adulthood. In these cases, people are usually diagnosed with “combined immunodeficiency (CID)” since signs and symptoms are initially milder than those seen in SCID. However, the immune response becomes more damaged the longer the condition goes undiagnosed.
A family history of unexplained infant death, or infant death caused by an infection, is an important clue leading to a possible diagnosis of SCID, particularly in families in whom the parents are biologically related.
In families where there has been a previous child with SCID, this may prompt the diagnosis in the newborn, even before symptoms have developed. In fact, SCID can be diagnosed before the baby is born, by comparing cells from a small amount of embryonic fluid, extracted during amniocentesis, to a previously analyzed sample from the older sibling diagnosed with the disorder. Results of tests or cell analyses from a sibling or siblings with SCID can be invaluable.
Children who present within the first 6 months of life are more likely to have SCID than other types of immunodeficiency disorders. In 85%-90% of patients with ADA-SCID, disease starts before the age of 1 year.3 The typical signs and symptoms are characterized by an excessive number of persistent or recurrent opportunistic infections and certain anatomical abnormalities. Infants with SCID may have neurological problems, such as developmental delay, movement disorders, and hearing loss. These infants also tend to have very small, underdeveloped thymus glands and abnormalities of the spleen, as well as the absence of lymph nodes and tonsils. These abnormalities can be diagnosed through direct examination, laboratory analyses, and radiographic screening.
In addition to the signs and symptoms typical with other types of SCID, ADA-SCID causes multiple skeletal abnormalities including chondro-osseous dysplasia (abnormal development of cartilage and bone), neurological defects involving motor function, partial or profound deafness, and hepatic (liver) dysfunction. These patients also show defects in cognitive and behavioral function. Infants with ADA-SCID have the lowest T-, B-, and NK-cell counts found in any type of SCID.
Diagnosis – Adenosine deaminase deficiency
Genetic testing is available for ADA, the gene known to cause adenosine deaminase deficiency. Carrier testing for at-risk relatives and prenatal testing are possible if the disease-causing mutations in the family are known.
In some states of the United States, newborns are screened for various conditions that can cause severe combined immunodeficiency, including adenosine deaminase deficiency (ADA). In the absence of newborn screening, ADA is typically suspected in people with frequent infections and other characteristic symptoms. The diagnosis can be confirmed by blood tests that demonstrates the following:
Evidence of combined immunodeficiency such as low levels of certain types of lymphocytes (white blood cells) and/or absent lymphoid tissue (i.e. thymus, tonsils, and lymph nodes)
Low (<1%) adenosine deaminase (the enzyme encoded by the ADA gene) activity
Other tests that are helpful in establishing a diagnosis include x-rays that show ADA-associated bone abnormalities and genetic testing that identifies a change (mutation) in each copy of the ADA gene.
Prognosis – Adenosine deaminase deficiency
The long-term outlook (prognosis) for people with adenosine deaminase deficiency (ADA) varies depending on the severity of the condition, the timing of the diagnosis and the response to treatment. For example, without early diagnosis and treatment, babies with ADA usually do not survive past age 2. However, if bone marrow transplant/stem cell transplant (transplantation of blood-forming stem cells from the bone marrow of a healthy person) is effective, the prognosis is typically good.
Inheritance
Adenosine deaminase deficiency is inherited in an autosomal recessive manner. This means that to be affected, a person must have a mutation in both copies of the responsible gene in each cell. The parents of an affected person usually each carry one mutated copy of the gene and are referred to as carriers. Carriers typically do not show signs or symptoms of the condition. When two carriers of an autosomal recessive condition have children, each child has a 25% (1 in 4) risk to have the condition, a 50% (1 in 2) risk to be a carrier like each of the parents, and a 25% chance to not have the condition and not be a carrier.
Treatment
The treatment of adenosine deaminase deficiency (ADA) usually consists of the following:
- Early diagnosis and therapy for bacterial, viral, and fungal infections
- Prophylactic medications for certain types of pneumonia
- Intravenous (IV) immunoglobulin to boost the body’s natural response to infections
It is also important to restore the function of the immune system, if possible. The most effective treatment is transplantation of blood-forming stem cells from the bone marrow of a healthy person (called bone marrow transplant/stem cell transplant or BMT/SCT). This therapy cures the condition in approximately 70% or more of people with severe combined immunodeficiency (SCID), including those affected by ADA. If a BMT/SCT is not an option, enzyme replacement therapy (ERT) may be recommended. ERT is a treatment that replaces the enzyme that is missing or not working properly (adenosine deaminase) with a synthetic form of the enzyme.
You’ll want to start treatment right away. Find a specialist who treats immune deficiencies.
Your doctor will prescribe antibiotic, antifungal, or antiviral medications to treat any existing infections.
Your doctor may also prescribe antibiotics to prevent new infections. A baby or child with ADA-SCID may need to spend some time in an isolated hospital room, but his parents will be able to be with him.
Although it doesn’t cure the disease, enzyme replacement therapy (ERT) may help your immune system work better and prevent infections. In this therapy, you get injections of healthy enzymes, usually from a cow.
The only way to cure ADA-SCID is with a stem cell transplant. Doctors will put healthy stem cells into your body to try to rebuild your immune system. It’s most successful in infants, and when the donor stem cells come from a close relative. In some cases, people need chemotherapy before getting a transplant to kill damaged cells first.
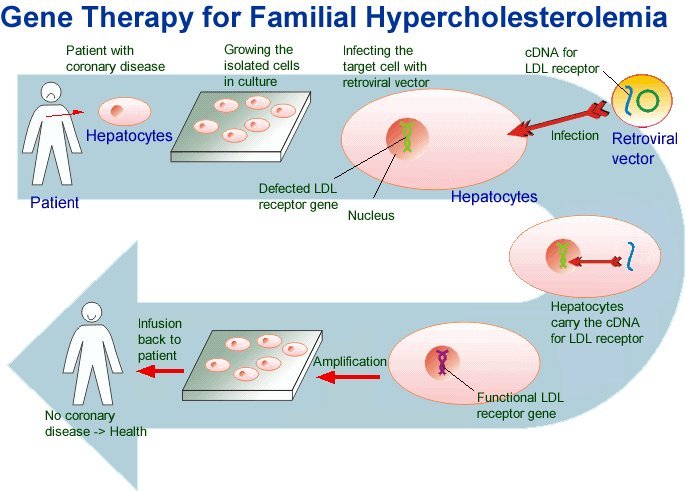
Scientists are looking for more ways to treat ADA-SCID. Studies using gene therapy show promise. In these studies, researchers add healthy genes to your own cells in a lab and transplant them back into your body to correct faulty cells.
Routine Screening for SCID
Not all states require routine screening for SCID (including ADA-SCID) in newborns. The test is routinely offered, but not yet required in some states, and in others the test is required, but not yet implemented.5 The Advisory Committee on Heritable Disorders in Newborns and Children has recommended routine newborn screening for SCID and will be determining whether national screening should be mandatory.6-7 Use of the newer technology known as TREC (T-cell Receptor Excision Circles) can be done at birth to identify babies with SCID and intervene with supportive care. Babies found to be positive can have assays (enzyme tests) for ADA to identify babies with ADA deficiency before symptoms or illness develop.
What are the risk factors for severe combined immunodeficiency?
There are risk factors of age, sex and ethnicity:
- Most patients with SCID present by age 3 months.
- The most common form of SCID (responsible for about 45% of all cases) is X-linked and only presents in males. Other forms, however, can present in males and females equally.
- Certain ethnic groups — including the Finnish, North Africans, Italians, Navajos and Apaches — appear to carry a greater risk for specific forms of this condition.
Known family history of the disease
Complete blood count (CBC) plus a manual differential to calculate the absolute lymphocyte count
T-cell function tests
In utero prenatal genetic testing via chorionic villous sampling (CVS) or amniocentesis
- Neonatal screening
Diagnosis
General: Severe combined immunodeficiency (SCID) is generally suspected in infants younger than one year old who suffer from frequent and/or serious infections (especially fungal infections that are resistant to treatment), persistent diarrhea, weight loss, and chronic (ongoing) skin infections. These patients may have an enlarged liver, spleen, and/or lymph nodes, which indicate that the body is fighting off an infection. A series of tests (listed below) are necessary to confirm a diagnosis.
Complete blood count (CBC): A complete blood count (CBC) can be conducted to determine the number of lymphocytes in the blood. Lymphocytes are special cells in the bloodstream that support the immune system and keep a patient healthy. The blood count can also show if other immune cells in the blood are absent or out of balance. During the procedure, a small sample of blood is taken and analyzed under the microscope. Decreased levels of lymphocytes may indicate the presence of SCID.
Blood smear: A blood smear is often used to measure the number of lymphocytes (a kind of white blood cell in the patient’s blood). The white blood cells, which are part of the body’s immune system, help fight against diseases and infections. Healthy patients typically have more than 1,500 lymphocytes per cubic millimeter of blood. Infants who have SCID usually have significantly decreased numbers of white blood cells.
Mitogen stimulation assay: Lymphocyte function can be tested by its reaction to specific stimulation with plant extracts called mitogens. Mitogens are added to a sample of the patient’s blood to stimulate mitosis (cell division) of lymphocytes. This allows the healthcare provider to observe the lymphocytes during mitosis. This test can detect abnormal or dysfunctional lymphocytes that cause SCID.
Immunoglobulin test: An immunoglobulin test measures the levels of antibodies (glycoprotein that detects and binds to foreign substances that enter the body) in the blood. Patients who have SCID will have extremely low levels of immunoglobulin, especially two types called immunoglobulin A (IgA) and immunoglobulin (IgM). However, soon after birth, immunoglobulin G (IgG) levels may still be high because the newborn still has healthy maternal IgG that it received through the umbilical cord before birth.
X-ray: An X-ray may detect an absent or abnormally small thymus gland, which is associated with SCID. Some T-cells are produced in the thymus gland. If the thymus gland is abnormally small, an insufficient amount of T-cells may be produced causing SCID. An X-ray may also detect inflammation of the lung caused by pulmonary infections like interstitial pneumonitis or pneumonia.
Prenatal testing: Amniocentesis, chorionic villous sampling, or cord blood can be used to diagnose a fetus with SCID. These procedures are most often conducted in patients who have a family history of SCID. Amniocentesis is performed at about 15-18 weeks of gestation. During the procedure, a long, thin needle is inserted into the pregnant woman’s abdominal wall to the uterus. A small amount of fluid is removed from the sac that surrounds the fetus. The fluid is then analyzed for genetic abnormalities. There is a risk of infection or injury to the fetus, and a chance of miscarriage.
During chronionic villus sampling (CVS), a small piece of tissue (chorionic villi) is removed from the uterus during early pregnancy to screen the fetus for genetic defects. Depending on where the placenta is located, CVS can be performed through the cervix (transcervical) or through the abdomen (transabdominal). The risks of infections or fetal damage are slightly higher than the risks of amniocentesis. Fetal loss occurs about two percent of the time.
During a cord blood test, a sample of blood is taken from the umbilical cord at the time of birth. The sample is then analyzed in a laboratory to determine whether the newborn has genetic abnormalities that indicate SCID.
Prevention
Because severe combined immunodeficiency (SCID) is a genetic disorder, there is currently no known method of prevention. However, individuals can be tested to determine whether they are carriers of the disease.
Preventative treatment against Pneumocystis jiroveci pneumonia (previously called Pneumocystis carinii pneumonia) is recommended for patients who are older than two months, until their T-cell function returns to normal after a bone marrow transplant. Trimethoprim-Sulfamethoxazole (Bactrim, Bactrim DS, Septra, Septra DS) is the standard drug of choice. Patients receive intravenous (IV) injections into their bloodstream three times a week.
For more information visit us our website: https://www.healthinfi.com
0 200

1 Comment