Accutane Embryopathy
Accutane embryopathy is a rare disorder and it is also called as Fetal Retinoid Syndrome. Accutane Embryopathy is a characteristic pattern of mental and physical birth defects that results from maternal use of retinoids, the synthetic vitamin Aderivative during pregnancy. The most well known retinoid is Isotretinoin (Accutane), a drug used to treat severe nodular acne. Birth defects associated with fetal retinoid syndrome include: hydrocephalus, microcephaly, intellectual disabilities, ear and eye abnormalities, cleft palate and other facial differences, and heart defects. Isotretinoin can cause these birth defects in the early weeks of pregnancy, even before a woman knows that she is pregnant.
Symptoms – Accutane embryopathya
Growth delays before and after birth (prenatal and postnatal groawth retardation)
- Malformations of the skull and facial (craniofacial) region
- Central nervous system abnormalities
- Heart abnormalities
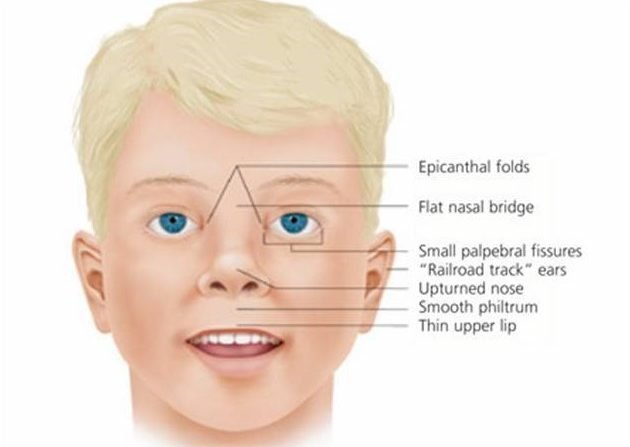
- Specific facial features (sharply sloping forehead, small jaw flattened bridge of the nose, and an abnormal size and/or placing of the eye sockets and eyes)
- Abnormal teeth mottling, facial nerve paralysis, and hair pattern abnormalities
- Malformed ears (external ear abnormalities vary from very small (Microtia) or missing ear (anotia), and stenosis of external canal)
- Tetralogy of fallot
- Conotruncal malformations
- Transposition of great cardiac vessels
- Double outlet right ventricle
- Aortic arch hypoplasia
- Microcephaly (small head)
- Hydrocephalus
Mild to moderate mental retardation with learning disabilities
- Cerebellar hypoplasia
- Thymic aplasia and thymic hypoplasia
- Cleft palate
Causes – Accutane embryopathy
- Hypertension
- Diabetic retinopathy
- Glaucoma
- Cholesterol emboli
- Papilledema
- Pigmented crescent
- Macular degeneration
- Retinal detachment
- Acute optic neuritis
- Optic atrophy
- Retinal hemorrhage
- Chorioretinal exudates
- Lipemia retinalis
- Central retinal artery occlusion
- Central retinal vein occlusion
- Angioid streaks
- Hyperviscosity Top
Maternal use of synthetic vitamin A (retinoids) such as isotretinoin (Accutane) during pregnancy can result in multiple effects on the developing embryo and fetus including miscarriage, premature delivery and a variety of birth defects. Additional retinoids include actiretin (Soriatane), etretinate (Tegison), and retinoin (Vesanoid).
A heightened risk of birth defects exists in women who become pregnant while taking retinoids such as Accutane. There is disagreement in the medical literature as to the specific risks present. One source in the medical literature estimated that a 35 percent risk of fetal retinoid syndrome exists in children of women who take Accutane beyond the 15th day following conception. Some researchers believe that birth defects do not occur in women who discontinue Accutane use one month before conception. It is also unknown what specific dosage of retinoids may result in birth defects. Some women taking low doses of retinoids have had children with severe symptoms of fetal retinoid syndrome. More research is necessary to determine the specific risks and long-term effects of taking retinoids such as isotretinoin (Accutane) during pregnancy.
The structures of the body most often affected in infants with fetal retinoid syndrome are the brain, the heart, and craniofacial structures especially the ears and palate.
Diagnosis – Accutane embryopathy
History of Accutane use by the mother during the first trimester of pregnancy
Recognition of the gross physical abnormalities in an exposed infant
A prenatal ultrasound evaluation may detect abnormalities such as heart defects, hydrocephalus or microcephaly, or some craniofacial abnormalities
Prognosis – Accutane embryopathy
Accutane embryopathy is a serious medical condition that is directly related to a maternal use of Accutane during the first trimester of her pregnancy
Although most individuals with Accutane embryopathy will have a normal lifespan, others may die at a young age due to complex internal abnormalities
Mild or moderate mental handicap is common even when there are no obvious physical features of Accutane embryopathy
Treatment – Accutane embryopathy
The care of an infant with Accutane embryopathy after delivery is primarily symptomatic
Infants with serious heart abnormalities will need to be evaluated by cardiologists and may require surgery
Infants with brain and nervous system abnormalities, such as hydrocephalus may require shunt placement soon after birth
Speech therapy and behavioral counseling is required
Signs & Symptoms
Characteristic features of infants with fetal retinoid syndrome include abnormalities of the head and face (craniofacial) region, central nervous system, and cardiovascular system. The specific symptoms and physical findings will vary from one infant to another. Affected infants will not have all of the symptoms listed below. For example, an affected infant may have below average intelligence and learning disabilities, but no major structural abnormalities; another infant may have serious medical complications.
Affected infants often display small, low-set ears (microtia) with narrowing (stenosis) of the ear canals. Abnormalities of the middle and inner ears may also be present. Additional craniofacial findings include widely spaced eyes (hypertelorism), incomplete closure of the roof of the mouth (cleft palate), an abnormal groove in the upper lip (cleft lip), and/or underdevelopment of the middle area of the face (midface hypoplasia). In some cases, affected infants may experience paralysis (palsy) of certain facial nerves. Affected infants may also have a condition known as microcephaly, which indicates that the head circumference is smaller than would be expected for age and sex.
Some infants with fetal retinoid syndrome will develop cardiovascular abnormalities including structural (anatomical) malformations of the heart including transposition of the great vessels; hypoplastic left heart syndrome; ventricular septal defects (VSDs); and a condition known as tetralogy of Fallot. Hypoplastic left heart syndrome is characterized by underdevelopment of the left ventricle, the aortic and/or mitral valves, and the ascending aorta.
VSDs are abnormal openings that may occur in any portion of the ventricular septum, the fibrous partition that divides the heart’s two lower chambers (ventricles). The size and location of the defect determine the severity of the symptoms. Small VSDs may close without treatment (spontaneously) or become less significant as affected children mature and grow. Due to moderately-sized defects, the heart may be unable to pump blood effectively (congestive heart failure), resulting in an abnormally rapid rate of breathing (tachypnea), wheezing, unusually fast heartbeat (tachycardia), and/or failure to grow and gain weight at the expected rate (failure to thrive). In some cases, without appropriate treatment, large VSDs may cause life-threatening complications during infancy.
Tetralogy of Fallot is a rare form of cyanotic heart disease. Cyanosis is abnormal bluish discoloration of the skin and mucous membranes that occurs due to low levels of circulating oxygen in the blood. Tetralogy of Fallot consists of a combination of four different heart defects: a ventricular septal defect; obstructed outflow of blood from the right ventricle to the lungs due to an abnormal narrowing of the opening between the pulmonary artery and the right ventricle of the heart (pulmonary stenosis); a displaced aorta that causes blood to flow into the aorta from both the right and left ventricles; and abnormal enlargement of the right ventricle.
Some infants with fetal retinoid syndrome may develop central nervous system abnormalities including a condition in which accumulation of excessive cerebrospinal fluid (CSF) in the skull causes pressure on the tissues of the brain, resulting in a variety of symptoms (hydrocephalus). Affected infants may also develop cysts in an area of the brain that houses the brainstem and cerebellum (posterior fossa). Infants with fetal retinoid syndrome may have below average intelligence, experience learning disabilities, and exhibit delays in reaching developmental milestones such as sitting or crawling. In some cases, the forebrain (prosencephalon) may fail to develop (holoprosencephaly).
Infants with fetal retinoid syndrome often experience abnormalities of thymus function. The thymus gland is located below the thyroid gland in the neck and front of the chest and is the primary gland of the lymphatic system, which is necessary for the normal functioning of the immune system. The parathyroid glands, located on the sides of the thyroid gland, are responsible for the maintenance of normal levels of calcium in the blood.
Additional abnormalities that may occur in some cases include webbing of the fingers (syndactyly), skeletal malformations affecting the legs and spines, and/or low muscle tone (hypotonia).
Affected Populations
Fetal retinoid syndrome affects males and females in equal numbers. The exact incidence of fetal retinoid syndrome is unknown and because many cases of fetal retinoid syndrome often go unrecognized, the disorder is under-diagnosed, making it difficult to determine the true frequency of the disorder in the general population.
According to the medical literature, women who are considering taking isotretinoin (Accutane) must first be given a pregnancy test approximately two weeks to a month before starting therapy. Woman on Accutane are encouraged to use two forms of contraception.
The guidelines for Accutane usage :
Isotretinoin is teratogenic, causes birth defects, and is Category X, meaning that there is no indication for its usage during pregnancy. In addition, it should not be taken while nursing.
Patients should take 2 pregnancy tests before starting isotretinoin and take a monthly pregnancy test while on the medication.
Two forms of birth control should be used starting 1 month before treatment, during the entire course of treatment, and for 1 month after ceasing treatment with isotretinoin. Even if one of the forms of contraception is hormonal, a second form should be used.
Providers should be knowledgeable about the use of isotretinoin and should read the educational materials and sign a letter of understanding before prescribing the medication. Yellow stickers are given to providers to affix to the prescriptions for isotretinoin. The yellow sticker indicates that the patient has met all criteria for receiving the medication, signed informed consent, and followed the pregnancy testing recommendations.
Pharmacists should dispense isotretinoin only when the yellow sticker is affixed to the prescription. Only a 30-day supply of medication should be dispensed at a time, to remind patients to take a pregnancy test monthly, before getting another 30-day supply.
Providers and consumers should realize that isotretinoin is a potent drug with the potential to cause birth defects and reserve its use for severe recalcitrant nodular acne. Isotretinoin is not for use in mild forms of acne and should not be shared among family members. Isotretinoin should not be purchased over the Internet without appropriate education and safeguards.
Demographics
The total number of women of reproductive age (15-44 years old) taking Accutane is unknown. However, since the 1990s, the overall number of prescriptions written for Accutane has increased over two hundred percent. Prescriptions are evenly divided between men and women, but women 30 years old or younger account for 80% of the patients among their sex.
A Dermatologic and Ophthalmic Drug Advisory Committee was convened at the FDA in September 2000. Patterns of Accutane use and the outcomes of Accutane-exposed pregnancies were presented at this meeting. Two overlapping sources of pregnancy data exist: one sponsored by the manufacturer of the drug, Roche Laboratories, and a second study maintained by the Slone Epidemiology Unit at the Boston University School of Public Health. Representatives from both institutions reviewed their outcome data up to that time. This data supports previous estimates of the frequency of AE.
A total of 1,995 exposed pregnancies have been reported between the years 1982 and 2000. These pregnancies have been voluntarily reported either directly to the manufacturer or to the Slone Survey. Although doctors have referred some, a majority of participating women obtained the appropriate phone numbers from the insert included with their medication. Elective terminations of pregnancy were performed in 1,214 pregnancies. Spontaneous pregnancy losses were reported in 213 pregnancies and 383 infants were delivered. Of these, 162, or 42%, were born with malformations consistent with AE.
The numbers from the Slone Survey, which began in 1989, represent a large subset of the data reported by Roche. Any woman to whom Accutane is prescribed is invited to contact and participate in the project. As of September 2000, the survey had identified a total of 1,019 pregnancies out of more than 300,000 women enrolled. Some women were already pregnant when they had started Accutane but others conceived while taking the drug. The pregnancy data allows for examination of the risk factors that lead to becoming pregnant as well as the pregnancy outcomes. Among the 1,019 pregnancies that occurred, 681 were electively terminated, 177 resulted in a spontaneous loss, and 117 infants were delivered. Only 60 of these infants were either examined or had medical records available to review. Eight of the 60 (13%) were diagnosed with AE. No information was available on the remaining 57 pregnancies.
Each couple in the general population has a background risk of 34% of having a child with any type of congenital birth defect. The medical literature has suggested a 25–35% risk of AE in infants exposed to Accutane prenatally. The combined Roche and Slone Survey data provided a risk of 42%. Although consistent with the medical literature, this slightly higher number probably reflects some bias in reporting. In other words, some mothers may report their pregnancy only after the birth of a child with AE. Normal births may go unreported. This type of retrospective analysis is not as helpful as prospective reporting in which pregnancies are enrolled before the outcome is known. To ensure objective reporting, the Slone Survey only enrolls their participants prospectively, ideally before the end of the first trimester of pregnancy. Even still, the Slone Survey estimates that it likely only has information on roughly 40% of all Accutane-exposed pregnancies.
Craniofacial
Malformed ears. Abnormalities of the ears, when present, involve both ears but may show different levels of severity ranging from mild external abnormalities to a very small or missing ear.
Underdevelopment of the skull and facial bones. This leads to a specific facial features including a sharply sloping forehead, small jaw (micrognathia), flattened bridge of the nose, and an abnormal size and/or placing of the eye sockets and eyes.
- Heart
- Structural defects, most of which require surgery to correct.
- Central nervous systerm
- Hydrocephalus , or abnormal accumulation of fluid within the brain. This is the most common type of brain abnormality and often is treated by placement of a shunt within the head to drain the fluid.
- Small head size (microcephaly)
- Structural or functional brain abnormalities
- Mild to moderate mental retardation or learning disabilities later in life. Either may be present even in the absence of physical abnormalities.
- Abnormal or very small thymus gland
- Cleft palate, or opening in the roof of the mouth
For more information visit us our website: https://www.healthinfi.com
2 500
1 Comment