ACD (Alveolar capillary dysplasia)
Alveolar capillary dysplasia with misalignment of pulmonary veins (ACD/MPV) is a disorder affecting the development of the lungs and their blood vessels. The disorder affects the millions of small air sacs (alveoli) in the lungs and the tiny blood vessels (capillaries) in the alveoli. It is through these alveolar capillaries that inhaled oxygen enters the bloodstream for distribution throughout the body and carbon dioxide leaves the bloodstream to be exhaled.
In ACD/MPV, the alveolar capillaries fail to develop normally. The number of capillaries is drastically reduced, and existing capillaries are improperly positioned within the walls of the alveoli. These abnormalities in capillary number and location impede the exchange of oxygen and carbon dioxide.
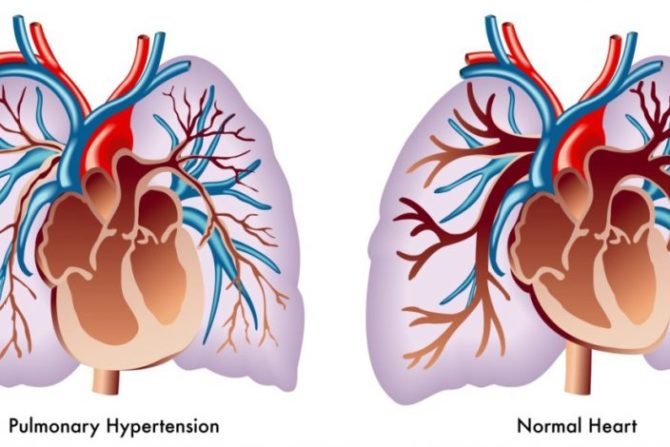
Other abnormalities of the blood vessels in the lungs also occur in ACD/MPV. The veins that carry blood from the lungs into the heart (pulmonary veins) are improperly positioned and may be abnormally bundled together with arteries that carry blood from the heart to the lungs (pulmonary arteries). The muscle tissue in the walls of the pulmonary arteries may be overgrown, resulting in thicker artery walls and a narrower channel. These changes restrict normal blood flow, which causes high blood pressure in the pulmonary arteries (pulmonary hypertension) and requires the heart to pump harder.
Alveolar capillary dysplasia with misalignment of the pulmonary veins (ACDMPV, OMIM# 265380) is a very rare disorder that is present at birth (congenital). Infants experience severe, life-threatening breathing problems and high blood pressure in the blood vessels of the lungs. These problems may occur within a few hours of birth. Almost all infants pass away within the first month of life. Very rarely, the disorder presents later (late-onset form). Infants often have additional symptoms including symptoms of the gastrointestinal tract, cardiovascular system and genitourinary system. In the vast majority of infants, AVDMPV is caused by changes (mutations) in the FOXF1 gene or in a loss of genetic material (deletion) that contains the FOXF1 gene or involves its distant regulatory genomic region (enhancer). The disorder is usually not inherited, but has in specific instances been seen to run in families.
Anemia of chronic disease (ACD) is second in frequency as a common cause of anemia, with iron deficiency anemia being the most frequent cause of anemia. In some clinical circumstances, such as in cancer clinics, ACD is the most common presentation. The chronic diseases most frequently associated with this type of anemia include chronic inflammatory disorders, chronic infections, neoplasms, and end-organ failure. Chronic inflammatory disorders can include diseases ranging from rheumatoid arthritis to inflammatory bowel disease. HIV-1 infections and other types of chronic infections are also known to be associated with this anemia. ACD also has an association with chronic liver disease, chronic kidney disease, and endocrinologic disorders. Typically, ACD occurs in older patients of both sexes with an apparent underlying chronic disease. However, age itself, especially after 85 years of age, is an indicator for this presentation.
When we breathe, air enters our lungs and fills tiny airsacs called alveoli. Each air sac is surrounded by an extensive capillary bed, which is a network of tiny, thin blood vessels. Capillaries are the smallest blood vessels in the body. In order for us to bring in oxygen and release carbon dioxide, the alveoli and capillaries must be properly aligned. If there are not enough capillaries or the capillaries are too far away from the surface of the alveoli, gas exchange cannot occur effectively, which means there is not enough oxygen for the body and the heart must work much harder.
Alveolar Capillary Dysplasia with misalignment of the pulmonary veins (ACDMPV) is a rare disorder where the microscopic capillaries around the air sacs fail to develop normally. There are fewer capillaries, and the ones that are present are not positioned correctly within the walls of the alveoli. This makes gas exchange (the delivery of oxygen from the lungs to the bloodstream) very difficult and ineffective.
Normally, the blood is brought to the capillaries by arteries, and the veins carry blood away from the alveoli to the heart in bundles with lymph vessels. In ACDMPV patients, these veins are found abnormally bundled with the arteries and the smooth muscle tissue in the arteries is often thickened, which restricts normal blood flow. When this occurs, it increases the blood pressure in the pulmonary arteries and makes the heart work harder. This causes pulmonary hypertension and is one of the main symptoms of this disease.
Differential Diagnosis
Idiopathic PPHN, which is generally distinguished from ACDMPV by its reversibility, most often resolving completely with pulmonary vasodilators, respiratory support, and/or ECMO;
Sepsis, pneumonia, and other infectious etiologies;
Primary respiratory disorders, including abnormalities of surfactant production (e.g., surfactant protein B deficiency, transporter abnormalities) or function (including hyaline membrane disease), pulmonary hypoplasia, congenital diaphragmatic hernia, and other rare diffuse interstitial lung disorders (e.g., acinar dysplasia, congenital alveolar dysplasia);
Congenital cardiopulmonary diseases associated with severe cyanosis and/or pulmonary hypertension, most notably pulmonary venous stenosis and total anomalous pulmonary venous return (however, it should be remembered that cardiac lesions have been reported in up to 25% of children with ACDMPV); and
Neurologic disorders, including perinatal asphyxia and congenital neuromuscular disorders.
Signs and Symptoms
ACDMPV is a pulmonary disease that presents in very early infancy. Infants generally become critically ill in the first days of life with severe hypoxemia and pulmonary hypertension, although limited reports have begun to emerge of presentation beyond the neonatal period in patients with atypical or late presenting ACDMPV. The majority of patients with ACDMPV (up to 80%) will have other associated anomalies of the cardiovascular, gastroinstestinal, urogenital, or musculoskeletal systems.
The initial presentation is identical to severe idiopathic pulmonary hypertension of the newborn. However, infants with ACDMPV do not respond, or respond only transiently to therapies that are usually effective in reversing this condition. Infants with ACDMPV do not improve despite maximal support in the intensive care nursery including mechanical ventilation, nitric oxide, and extracorporeal membrane oxygenation (ECMO).
Etiology/Epidemiology
The ACD Association has grown to close to 200 registered families from around the world and there are more than 100 cases of ACDMPV reported in medical literature worldwide. The incidence or prevalence of ACDMPV is not yet known, but there are clearly more cases than those that have been reported formally in the literature. For several reasons, these case reports almost certainly underestimate the true prevalence of ACDMPV. First, the definitive diagnosis of ACDMPV currently depends on histological examination of lung tissue on autopsy or ante mortem lung biopsy, and neither of these diagnostic modalities is universally pursued in the setting of a critically ill or dying newborn. Second, many infants born with ACDMPV have associated malformations in other organ systems that may allow the lung pathology to go undetected. Third, the diagnosis may be missed by pathologists unfamiliar with the disorder. It seems likely that some cases originally classified as idiopathic persistent pulmonary hypertension of the newborn (PPHN) may actually have been ACDMPV. Last, evidence is mounting to suggest that a less severe phenotype compatible with prolonged survival might exist, although definitive diagnostic criteria for this group are yet to be established.
No geographic pattern is apparent; cases have been distributed worldwide. More than 90% of affected infants are born at term, and in greater than 60% of cases, onset of cyanosis and respiratory failure occurs within 48 hours of birth. Most of the cases have been sporadic, de novo events, but approximately 10% involved siblings, suggesting a heritable form of the disease. For additional information, please refer to the information on ACDMPV genetics HERE.
Diagnosis
The diagnosis of ACDMPV should be considered in infants who present with severe hypoxemia and idiopathic pulmonary hypertension, and who do not respond appropriately after 7 to 10 days of neonatal intensive care treatment as described below. The majority of patients with ACDMPV (up to 80%) will have other associated anomalies of the cardiovascular, gastrointestinal, urogenital, or musculoskeletal systems. The initial chest radiograph is usually normal. If a cardiac catheterization is performed, there may be absence of the capillary blush phase.
The diagnosis can only be confirmed by lung biopsy or autopsy. Consulting a pathologist with experience in making this diagnosis may be helpful. Pathological features include a paucity of alveolar capillaries, widened alveolar septae, and increased muscularization of pulmonary arterioles. There is usually malpositioning (“misalignment”) of pulmonary veins in the bronchovascular bundle, but this is not required for the diagnosis. A focal distribution of disease has been described, which makes it necessary to examine multiple lung sections if ACDMPV is suspected.
Treatment
Standard Therapy
Standard therapies include mechanical ventilation, high concentrations of inspired oxygen, inhalational nitric oxide and ECMO support. These therapies prolong life by days to weeks, but have not led to long-term survival.
Investigational Therapy
Because none of the supportive therapies described above has changed the expected mortality due to ACDMPV, lung transplantation is currently the only option that might prolong survival. Achieving early diagnosis by lung biopsy is an essential first step, followed by strategies that sufficiently prolong survival. Lung transplantation may provide effective treatment for carefully selected affected infants. Donor availability continues to limit the utilization of lung transplantation for neonatal diseases.
Several patients have survived with atypical or late presenting ACDMPV long enough to receive lung transplants. According to a 2013 case series conducted by St. Louis Children’s Hospital (the most active pediatric lung transplant program in the world), four ACDMPV patients (ages 4 months, 5 months, 9 months and 20 months of age at time of transplant) with atypical presentations of ACDMPV each underwent a successful bilateral lung transplantation (BLT). As stated in the case study, “If they survive to BLT, patients with ACDMPV can have successful outcomes” and the ACDMPV patients “are alive at last follow-up at 1, 8, 9 and 12 years of age” (as of May 2013). In addition, in 2016, an ACDMPV patient in the UK and a second ACDMPV patient at Children’s Hospital of Philadelphia, Pennsylvania, USA each received a successful bilateral lung transplant at the ages of 5 months and 4 months, respectively.
Late Presenters and Long-Term Survivors
Reports of infants presenting with clinical features of ACDMPV beyond the neonatal period have begun to emerge. With such limited information, it is difficult to estimate the number of patients with delayed presentation of ACDMPV. Until more specific diagnostic tools become available, similarities in lung pathology and clinical findings should increase diagnosis using the same approach to lung tissue sampling in late presenters as for those who present early.
Genetic Changes
ACD/MPV can be caused by mutations in the FOXF1 gene. The protein produced from the FOXF1 gene is a transcription factor, which means that it attaches (binds) to specific regions of DNA and helps control the activity of many other genes. The FOXF1 protein is important in development of the lungs and their blood vessels. The FOXF1 protein is also involved in the development of the gastrointestinal tract. Mutations in the FOXF1 gene that cause ACD/MPV result in an inactive protein that cannot regulate development, leading to abnormal formation of the pulmonary blood vessels and gastrointestinal tract.
ACD/MPV can also be caused by a deletion of genetic material on the long arm of chromosome 16 in a region known as 16q24.1. This region includes several genes, including the FOXF1 gene. Deletion of one copy of the FOXF1 gene in each cell reduces the production of the FOXF1 protein. A shortage of FOXF1 protein affects the development of pulmonary blood vessels and causes the main features of ACD/MPV. Researchers suggest that the loss of other genes in this region probably causes the additional abnormalities, such as heart defects, seen in some infants with this disorder. Like FOXF1, these genes also provide instructions for making transcription factors that regulate development of various body systems before birth.
ACD/MPV is usually not inherited, and most affected people have no history of the disorder in their family. The genetic changes associated with this condition usually occur during the formation of reproductive cells (eggs and sperm) or in early fetal development. When the condition is caused by a FOXF1 gene mutation or deletion, one altered or missing gene in each cell is sufficient to cause the disorder. Individuals with ACD/MPV do not pass the genetic change on to their children because they do not live long enough to reproduce.
A few families have been identified in which more than one sibling has ACD/MPV. It is not clear how ACD/MPV is inherited in these families because no genetic changes have been identified.
Related Disorders
Symptoms of the following disorders can be similar to those of ACDMPV. Comparisons may be useful for a differential diagnosis.
Idiopathic persistent pulmonary hypertension of the newborn (PPHN) is characterized by markedly high blood pressure in the pulmonary artery (pulmonary hypertension) of infants that, in turn, causes blood to bypass its normal route and results in less oxygen than required being delivered to the lungs (hypoxemia). PPHN may occur without known cause (idiopathic), or it may arise in connection with many different neonatal cardiorespiratory disorders, including asphyxia, meconium aspiration syndrome (MAS), respiratory distress syndrome (RDS), and congenital diaphragmatic hernia. During fetal life, pulmonary blood flow is low, with less than 10% of the combined cardiac output directed to the lungs. Following birth, pulmonary vascular resistance falls dramatically as the lungs assume the function of gas exchange. In some newborn infants, the normal decrease in pulmonary vascular tone does not occur, resulting in persistent pulmonary hypertension of the newborn (PPHN). This syndrome results in substantial morbidity and mortality in otherwise healthy term infants. Most infants with PPHN respond to therapies such as inhaled nitric oxide and extracorporeal membrane oxygenation. Most (~90%) recover by the second week of life and survive.
Surfactant protein B deficiency is a congenital (present at birth and associated with a gene defect) disorder; of the lungs that occurs primarily in infants born prematurely and is a result of immaturity of the lungs. The mature lung contains a foamy fluid known as surfactant, a substance essential to expansion of the alveoli or air sacs of the lungs. Because of their immaturity, premature babies tend to lack surfactant. Without surfactant, the lungs cannot inflate, resulting in RDS. Pulmonary surfactant is a complex compound composed primarily of fatty substances and lesser amounts of cholesterol and surfactant-associated protein. When the protein component is missing, the surfactant cannot function properly. The surfactant, when functioning properly, lowers surface tension at the air-liquid interface in the alveoli of the lung, permitting the exchange of oxygen.
What are the symptoms?
You may find that ACD causes mild symptoms or no symptoms at all.
If you do have symptoms, you may feel dizzy, tired, and weak. You may also feel your heart pounding or feel short of breath. It may be hard to focus and think clearly.
How is it treated?
ACD is most often treated by treating the health problem that caused it. For example, treating rheumatoid arthritis can lower inflammation, which can then improve ACD. For ACD caused by cancer or chronic kidney disease, medicine can help the body make more red blood cells. These medicines are called erythropoietin stimulating agents, or ESAs. Severe anemia is treated with a blood transfusion of red blood cells, no matter what the cause is.
Only take iron if your doctor tells you to. Unless you also have iron deficiency anemia, taking iron does not help with ACD. If your iron level is normal, taking extra iron can be dangerous.
For more information visit us our website: https://www.healthinfi.com
0 200
No Comments